The Patria vaccine against Covid-19, the project in which the government of Mexico placed its hopes to stop depending on foreigners, it appears to be a very slow development and could even be ready until June 2022 and not in December as anticipated.
Whilehe initial 150 million pesos project promoted by the National Council of Science and Technology (Conacyt) seems stagnant, the government of President Andrés Manuel López Obrador has spent around 38 billion pesos on the purchase of vaccines abroad, while the deaths from Covid-19 are already around 254 thousand and total infections add up to 3.5 million since March 2019.
According to health authorities, the country faces its third wave of coronavirus infections and has experienced record numbers in the number of infections in recent weeks, such as that of August 12, when 24,975 new cases were recorded in one day.
The goal of Mexico being able to produce its own vaccine against Covid-19 It was to save in principle up to 855% compared to the acquisition of vaccines abroad.
Read: They will recruit volunteers in CDMX for phase 1 of the Patria vaccine
“After the success obtained in the preclinical tests and once the tests in humans are passed, we hope to have a 100% Mexican vaccine development against Covid-19 to protect the population and what will free us from depending on vaccines against Covid-19 produced abroad and will contribute to the strengthening of scientific sovereignty ”, promised Conacyt in April 2021.
However, that desire has soon been diluted with respect to the impact of Covid-19 in Mexico that has motivated the purchase of vaccines abroad and to which must be added the presidential instruction to inoculate, with at least one dose, all Mexicans by October.
According to the Ministry of Foreign Relations, Until August of this year, Mexico has bought 243 million 930 thousand doses- of which it has received 96 million 573 thousand 385 doses- and for which it has invested around 38 billion pesos, according to information from the Ministry of Finance and Public Credit (SHCP).
The vaccines that Mexico has received are from the pharmaceutical companies AstraZeneca, Pfizer, CanSino, Sputnik V and Sinovac, which also include those acquired through the United Nations (UN) Covax system.

Health authorities have applied 81.3 million doses of various vaccines achieving that 31.2 million people have the complete scheme.
Although María Elena Álvarez-Buylla, director of Conacyt set the month of December as the goal so that the Patria vaccine could have its emergency use permit by the Federal Commission for Protection against Sanitary Risks (Cofepris), the truth is that until May Only the beginning of volunteer recruitment has been reported.
“In Mexico City they will all be recruited and this vaccine development will be injected during the next days and weeks and we hope to have the results by the end of May,” Álvarez-Buylla commented at the morning conference on April 13, 2021.
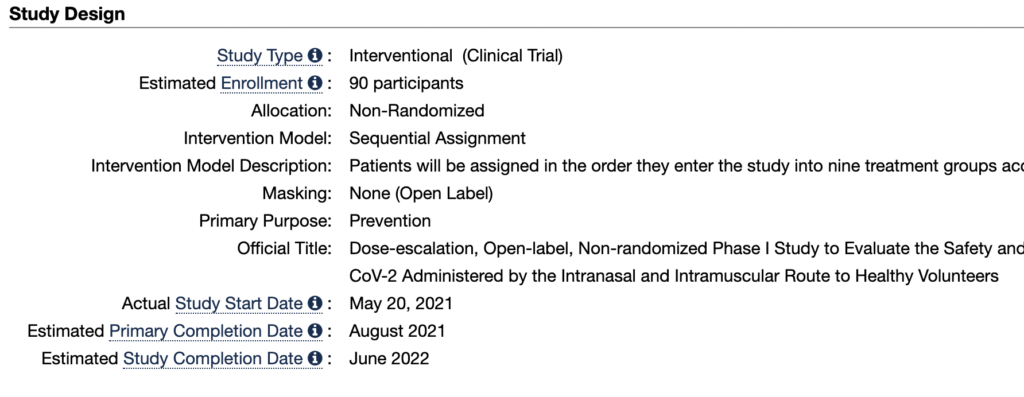
According to the website medicaltrials.gov, Until May 27, the phase of the analysis of the Patria vaccine in Mexico is recruitment and it is expected that it will be in August when the observation of the last volunteer patient in phase 1 will conclude.
However, according to estimates by Conacyt On April 13 of this year, between August and October, the last phase of the study should have started, that is, the 3.

The phase 1 clinical trial, which is directed by the Avimex laboratory, in collaboration with Conacyt and Amexid, is aimed at 90 volunteers from Mexico City at the Hospital Médica Sur and is directed by Dr. Samuel Ponce de León Rosales, specialist in infectious diseases of the UNAM.
“This is a phase 1, open-label, non-randomized, dose-escalation study using three doses and two schedules of administration of a recombinant SARS-CoV-2 vaccine based on a viral vector (Newcastle disease virus ) in 90 healthy volunteers at a single research site in Mexico City, ”says Avimex.
According to laboratory estimates, this clinical trial phase could yield conclusions by June 2022, that is, six months after the goal established by the Mexican government.
It should be noted that the information available on medicaltrails.gov does not specify the timing of phases 2 and 3 of the study.
Wanted for Forbes MexicoPonce León Rosales indicated that his team is focused on doing its job, but declined to comment on the progress of the Patria vaccine.
“Now we are really basically committed to carrying out our activity and at the moment we are not going to make any statement. I do not know based on what they say is late if there is no published calendar, “said the specialist.
This publishing house also looked for versions of the Conacyt, the Mexican Institute of Social Security (IMSS) and the Ministry of Health, but until the moment of publication of this note, no response was obtained to the proposal of the delay in the government forecasts.
It may interest you: Ebrard seeks cooperation from Russia for Mexican vaccine Patria
The silence on the advances in the clinical trials of the Patria vaccine have reached the Congress of the Union.
On July 13, the Permanent Commission asked the National Council of Science and Technology for a report on the research protocols and the results obtained regarding the development of the Patria vaccine.
At the international level, the study of the recombinant Newcastle disease paramyxovirus (rNDV), on which the Patria vaccine is based, is being conducted by scientists such as Dr. Constantino II López Macías, Sciences / Immunology at UNAM and the University of Zurich ; Peter Palese, Chemistry / Microbiology University of Vienna and Adolfo García Lastre, University of Salamanca and the Monte Sinai research center in New York.
* This note was originally published on August 29, 2021.
Follow us on Google News to stay always informed