On September 15 the European Parliament voted against a resolution motion of the Committee on the Environment, Public Health and Food Safety (ENVI), regarding the use of antibiotics in animals. Which, intended to expand antibiotic restrictions in animals, denying their access to all critical antimicrobials of greatest need.
What was the ENVI proposal about?
Specifically, the ENVI proposal opposed the delegated act of the European Commission that complements Regulation (EU) 2019/6. Which entered into force in 2022, establishing the designation criteria of antibiotics that should be reserved for humans.
This act already reflects the antimicrobials that will be reserved exclusively for human beings based on criteria that have had the scientific support of the European Medicines Agency, the European Food Safety Authority, the World Organization for Animal Health and the World Organization for Health (WHO).
However, the motion considered the Commission measures and insufficient science, and included a ban on the use in animals of all of the WHO’s top-priority critical antimicrobials: colistin, macrolides, third- and fourth-generation fluoroquinolones, as well as cephalosporins.
In fact, the European Platform for the Responsible Use of Medicines in Animals (Epruma). Ensures that ban without scientific criteria veterinary antibiotics do not even have to effectively combat the antimicrobial resistance and can even increase them. Because the pressure on the rest of the antibiotics that can be used would be greater.
The veterinary sector feared that these measures could damage health and welfare of the animals, and the OCV itself warned that it would have caused “irreparable damage to animal health and would cause a unnecessary suffering to animalsboth on farms and in homes.
The EMA and the European Commission have published the long-awaited opinion
Specifically, Parliament defeated the motion by 450 votes against against 204 in favor, something that was celebrated from the veterinary sector. What from the first moment was against the drastic restriction of antibiotics that was proposed.
And it is that, from the Spanish Veterinary College Organization (OCV), through the European Federation of Veterinarians (FVE). And even the Commissioner for Health and Food Safety, Stella Kyriakides, were against expanding the use of antibiotics in animals.
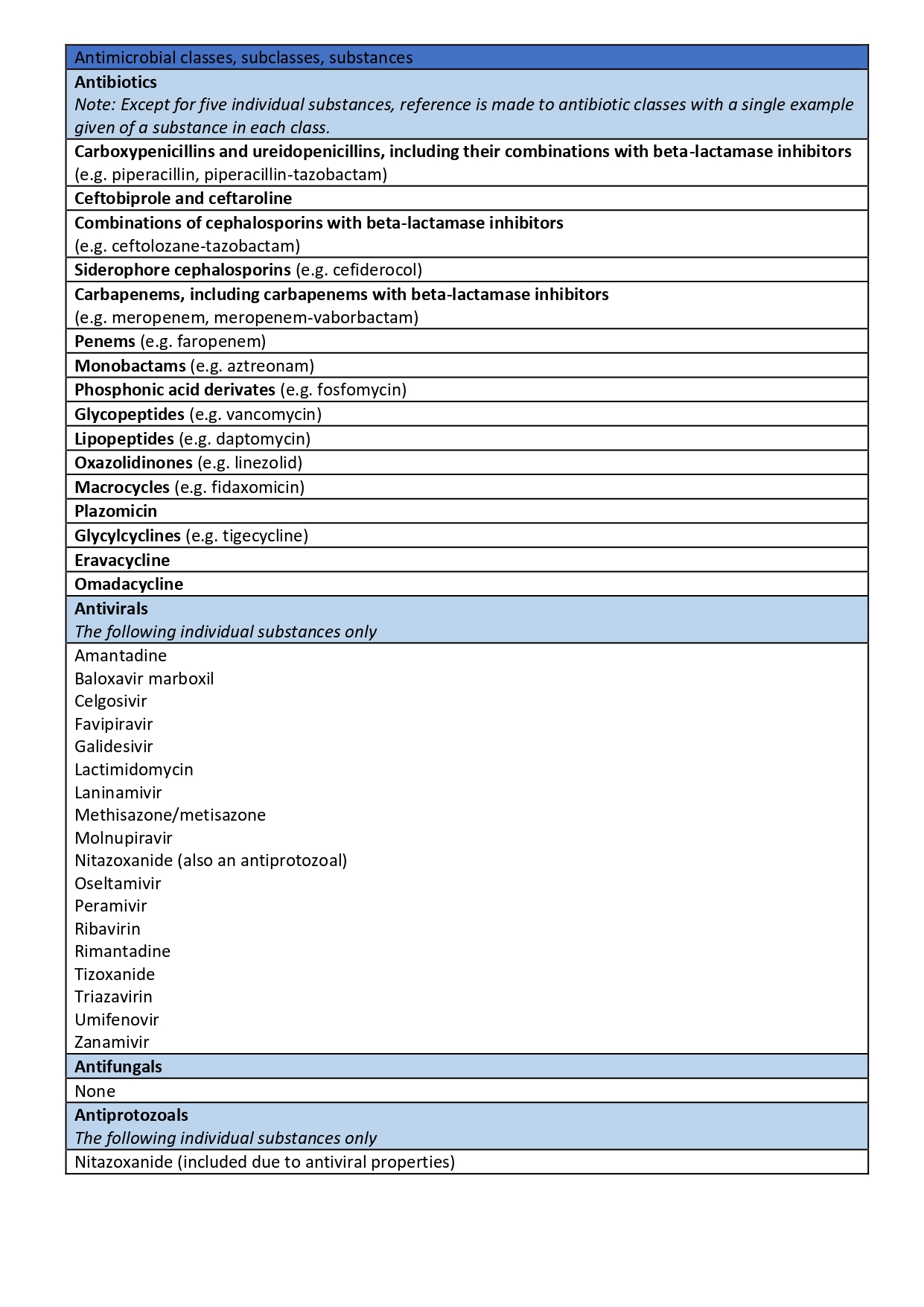
Now, the European Medicines Agency (EMA) and the European Commission have published the long-awaited opinion. This, about the antimicrobial designation or groups of antimicrobials reserved for the treatment of certain infections in humans.
After knowing the list of antimicrobials, the FVE has applauded “the scientifically based evaluation carried out, which fully applies the ‘One Health’ approach”. “This opinion confirms that Europe remains firmly anchored in a holistic and evidence-based approach to ensuring health. As well as the well-being of humans and animals in Europe”, the European veterinarians have celebrated.
Likewise, the FVE has highlighted that “the opinion safeguards the availability of all important substances for the veterinary Medicineincluding third and fourth generation cephalosporins, polymyxins and colistin, macrolides and fluoroquinolones”.
Here is the list of veterinary antibiotics reserved for humans approved by the EMA:
Medical examiner; key piece in the penal system that is increasing
The BEST Orthopedics and Traumatology hospitals in Mexico
Conamed launches course for the prevention of conflict in medical care